These results reveal that it is possible to tune the emission color of these compounds by changing the chemical environment of the $SbO_4$ matrix.
Lanthanide-containing materials comprise a wide range of scientifically and technologically important compounds. They are chemically designed and produced by using different routes depending on the final target: single crystals, glasses, organic-inorganic hybrids, and ceramics. A huge variety of properties can be obtained depending on the choice of the lanthanide, the host matrices and crystalline structures in which they are inserted, either as a dopant or as self-activated element. Known for possessing rich luminescence properties, lanthanides-containing materials have been used in many technological applications, such as laser materials, flat panel displays, cathode ray tubes, up-conversion devices, white-light emitting diodes, X-Ray scintillators, phosphors, and emitters.
Lanthanide-based materials emitting multiple visible colors have become an attractive research focus because of their important role in the field of light display, lasers and optoelectronic devices. Recently, Kisla P.F. Siqueira et al. (1) have reported on the complete series of $Ln \rm SbO_4$ (excepted cerium and promethium): lanthanide-containing ceramics emitting various visible colors were achieved by tuning the chemical environment and temperature into the antimony matrices. Their crystalline structures were resolved through conventional XRD and high-resolution synchrotron X-Ray diffraction.
Two different arrangements belonging to the monoclinic structure were identified and related to the ionic radius of the lanthanide metal. Orthoantimonates with largest lanthanide ions (La and Pr) belong to the $ \rm P2_1/n$ space group, while compositions involving intermediate and smallest lanthanides (Nd-Lu) were better described under the $ \rm P2_1/c$ setting. Vibrational features were investigated by Raman spectroscopy, which allow to determine the characteristic phonons for each structure and, consequently, to establish the relationship between chemical environment and vibrational modes.
Representative compositions for this orthoantimonate series ($\rm SmSbO_4$ and $\rm TbSbO_4$) were thoroughly explored in terms of their optical properties as a function of temperature under ultraviolet radiation. The results showed that these materials present strong orange (Sm) and green (Tb) emissions.
Synthesis and Structural Properties
Synthesis of the $Ln \rm SbO_4$ materials was obtained through solid-state reactions by using $Ln_2 \rm O_3$, $\rm Pr_6O_{11}$, $\rm Tb_4O_7$, and $\rm Sb_2O_5$ as starting materials.
All samples were firstly characterized by conventional X-Ray diffraction: high resolution synchrotron diffraction measurements were taken at room temperature at the superconducting wiggler XDS beamline of the Brazilian Synchrotron Light Source (LNLS) at λ= 0.65319 Å. Representative SXRD profiles of $Ln\rm SbO_4$ for Ln=La and Ln=Eu are given in Figures 1a and 1b, respectively. The two profiles are very different, indicating distinct crystal arrangements. For both materials a good agreement between observed and calculated diffraction data were obtained using monoclinic structures.
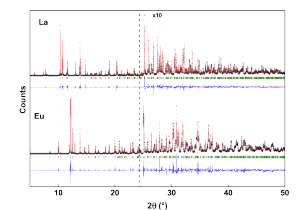
Figure 1. SXRD patterns of $Ln \rm SbO_4$ for Ln=La and Ln =Eu at λ= 0.65319 Å. The cross symbols and solid lines represent observed and calculated patterns, respectively. The difference curves are shown at the bottom of each figure; Vertical bars indicate expected Bragg positions according to the crystal structure models described in the text.
The La-based compound is isostructural to $\rm \beta-PrSbO_4 (P2_1/n)$, while the Eu one is similar to $\rm \alpha-PrSbO_4 (P2_1/c)$. An extensive investigation of the room-temperature Raman phonons and vibrational properties for all $Ln \rm SbO_4$ compounds was carried out for the first time. The results from Raman scattering indicated two different fingerprints in agreement with the crystal structures observed by SXRD.
Optical Properties
The optical properties of self-activated $Ln\rm SbO_4$ materials were investigated aiming to look for new luminescent emitters. For these compounds, luminescence is an inherent feature and this phenomenon occurs due to their chemical nature, i.e., no dopants are added: $\rm SmSbO_4$ and $\rm TbSbO_4$ were studied extensively, in particular the various types of energy transitions, at different temperatures. It is well known that the thermal quenching (reduction in luminescence intensity with increasing temperature, due to the opening up of competing non-radiative relaxation pathways) behavior is an important feature on evaluating the characteristics of luminescent materials. In $\rm SmSbO_4$, one of the types of energy transitions, the charge transfer (also called Charge Transfer Band-CTB) shows a strong thermal quenching: a factor 100 between 25 K and 275 K.
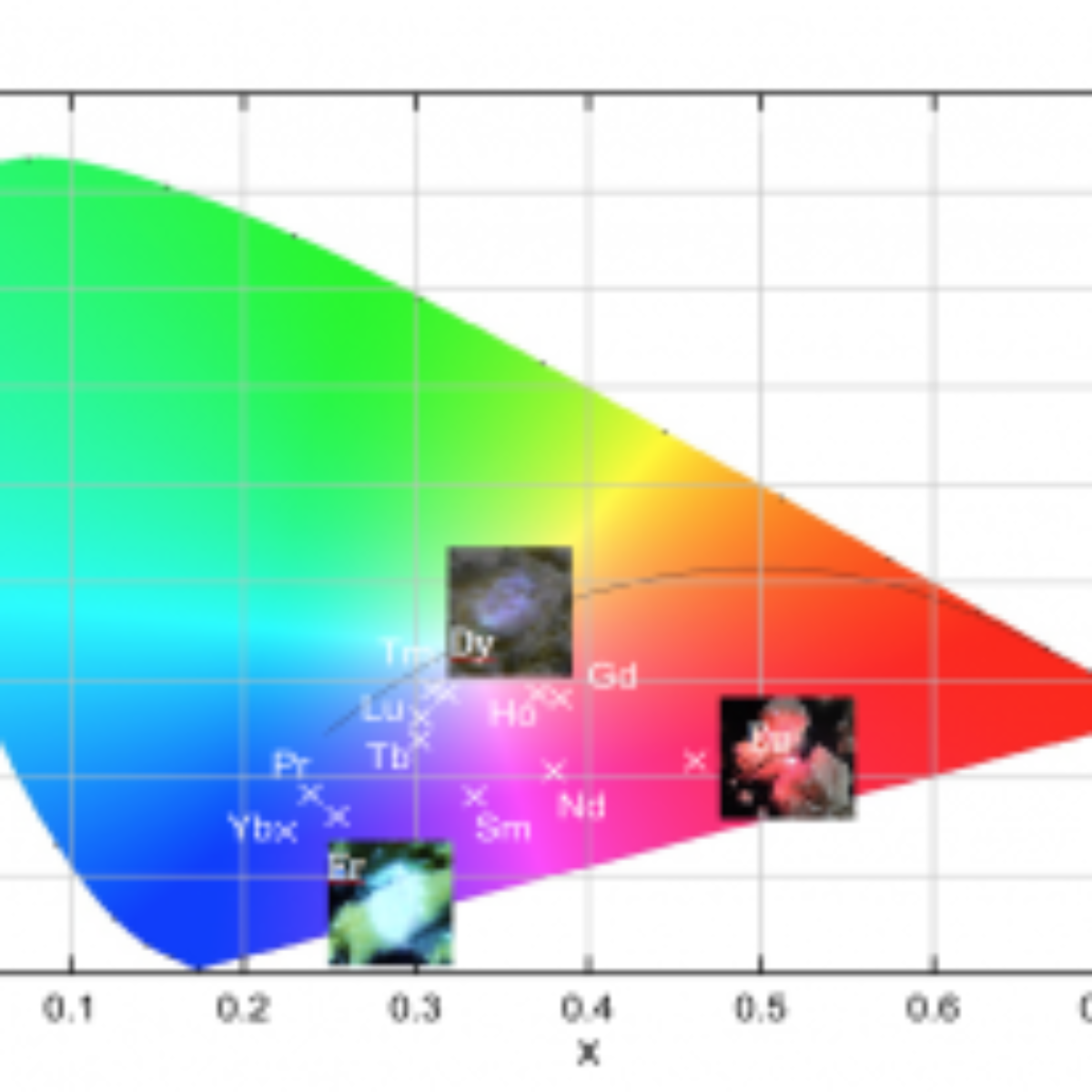
Color coordinates is another important merit factor for emitters. The color coordinates for the series of lanthanide orthoantimonates were calculated using the intensity-calibrated emission spectra data and are represented in a special plot: 1931 CIE color space chromaticity diagram (Figure 2). These results reveal that it is possible to tune the emission color of these compounds by changing the chemical environment of the $SbO_4$ matrix. The Figure 2 also presents the images for $\rm EuSbO_4$, $\rm DySbO_4$ and $\rm ErSbO_4$ ceramics in the moment that they are excited and emitting their respective colors as indicated at the chromatic diagram. All these results show that these materials are promising emitters due to the fact that the visible colors could be adjusted by the chemical environment of the lanthanide-containing $Ln \rm SbO_4$.
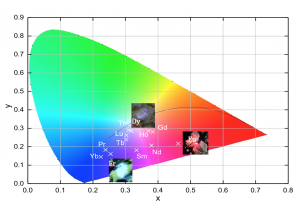
Figure 2. CIE chromaticity diagram coordinates of all $LnSbO_4$ compounds synthetized in this work.
Source:
(1) Kisla P. F. Siqueira, Patricia P. Lima, Rute A. S. Ferreira, Luis D. Carlos, Eduardo Bittar, Eduardo Granado, Juan Carlos González, Arturo Abelenda, Roberto L. Moreira and Anderson Dias, “Lanthanide Orthoantimonate Light Emitters: Structural, Vibrational and Optical Properties”, Chem. Mater., 2014, 26 (22), pp 6351–6360. DOI: 10.1021/cm502504b
Researchers at LNLS selected silver nanoparticles of different sizes and evaluated the effect of these nanoparticles on different bacterial strains.