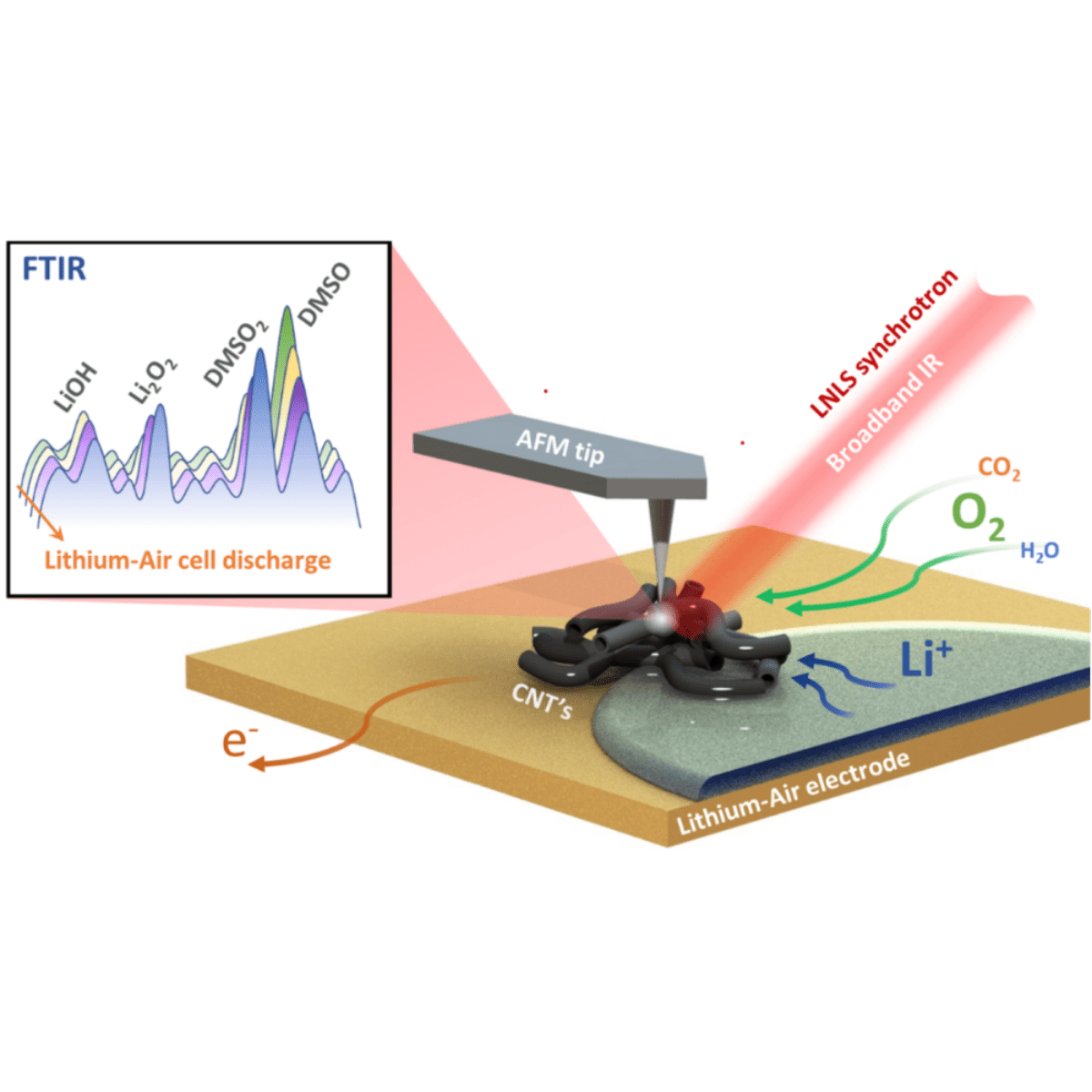
Lithium-air battery air electrode, consisting of gold substrate coated with carbon nanotubes (CNT), being analyzed by SINS.
Paper presents multiscale (nano-micro) FTIR analysis applied to the investigation of the chemical processes in Li-Air batteries
The growing demand for renewable energy sources underscores the urgency of developing new energy storage devices. One example is metal-air batteries, which could be the key to large-scale energy storage. This type of battery is a promising technology for commercial energy storage, as it offers about 10 times the energy density of Li-ion batteries, current market leaders.
Li-O2 or Li-Air batteries, as they are also called, operate with a lithium electrode and an air electrode, separated by a membrane embedded with conductive electrolyte and containing lithium ions. In the air electrode, during the energy supply stage, called discharge, products of the reduction of oxygen with Li+ are formed, such as Li2O2, Li2O, LiO2, which vary mainly according to the type of electrolyte, chemical composition and the structure of the air electrode. However, undesirable sub-products such as Li carbonates and hydroxides may also be formed when exposed to a real atmosphere (air).
Thus, despite being a promising technology to be applied commercially, there are several challenges to be overcome such as its operation under ambient conditions and the selection of an electrolyte that does not promote the formation of undesirable subproducts. In this context, understanding how carbon dioxide and moisture present in the atmosphere affect the chemical processes of the battery is essential. Furthermore, understanding the action of the electrolyte during the battery operation process can lead to the construction of more efficient devices. The knowledge of the parallel reactions that occur in the battery is also essential to optimize and develop them to provide more energy and to increase their lifetime. In addition, the development of air electrodes is another critical step to improve the performance of the devices and the multiscale analysis of the electrodes. The reactions at the electrode-electrolyte interface can provide valuable information in this process.
In this direction, researchers from the State University of Campinas (UNICAMP), Brazil, in partnership with researchers from CNPEM, investigated the chemistry on the surface of carbon-based air electrodes during battery discharge. With multiscale analyses carried out at the Imbuia beamline at the Sirius synchrotron light source of the Brazilian Synchrotron Light Laboratory (LNLS), the work was the cover of the journal Advanced Energy Materials and highlights the applicability of this technique in the study of batteries.
Using micro-FTIR and synchrotron infrared nanospectroscopy (SINS) techniques, it was possible to show that the chemical reactions studied at microscale can be extended to nanoscale in Li-Air batteries. The results showed that the reaction rate does not interfere in the chemical composition of the discharge products, with a homogeneous distribution of the products in nano- and microscale. In addition, due to the high sensitivity, the techniques allowed to understand the degradation of electrolytes in real time under operating conditions was highlighted.
Source: [1] Thayane C. M. Nepel, Chayene G. Anchieta, Leticia F. Cremasco, Bianca P. Sousa, André N. Miranda, Lorrane C. C. B. Oliveira, Bruno A. B. Francisco, Julia P. de O. Júlio, Francisco C. B. Maia, Raul O. Freitas, Cristiane B. Rodella, Rubens M. Filho e Gustavo Doubek. In Situ Infrared Micro and Nanospectroscopy for Discharge Chemical Composition Investigation of Non-Aqueous Lithium–Air Cells, Advanced Energy Materials 22 (2021) 101884. DOI: 10.1002/aenm.202101884.
Paper presents microfabrication and characterization of a multifunctional device suitable for the combination of X-ray analytical techniques
A prototype developed at CNPEM used sophisticated resources to assess device efficiency