Paper published at Science Magazine investigates unexpected molecular interactions that affect cell function and could cause disease
Cells conduct a series of interconnected biochemical reactions to obtain energy and respond to infections and different stressors. These reactions comprise what are known as metabolic pathways, which interact in complex networks and regulate various cellular processes. Understanding how these networks interconnect and work to regulate cellular processes is a challenge, because this signaling often depends on interactions between proteins and small molecules known as metabolites that involve low-affinity molecular bonds which are extremely difficult to identify.
Researchers from Brazil, the United States, China, South Korea, England, and Germany joined forces to investigate how protein-metabolite interactions (PMIs) affect carbohydrate metabolism pathways. Their findings have just been published in the respected journal Science. This high-impact article includes data obtained at Sirius, the cutting-edge synchrotron light source planned and built by the Brazilian Center for Research in Energy and Materials (CNPEM), an organization overseen by the Ministry of Science, Technology and Innovation (MCTI).
The article describes results obtained from a new technology known as MIDAS (mass spectrometry integrated with equilibrium dialysis for the discovery of allostery systematically), proposed by researchers at the University of Utah and tested by groups in different countries to investigate the subtle interactions between proteins and metabolites that may regulate countless cellular processes.
Using MIDAS, the researchers analyzed interactions between 33 enzymes of human carbohydrate metabolism and 401 metabolites, identifying 830 molecular PMIs. Although some of the identified PMIs involved previously known regulators, substrates, and products, this approach also made it possible for the researchers to discover many previously unknown PMIs from various metabolic pathways, including fat and carbohydrate metabolism mechanisms in different types of tissue that could be therapeutically used to block aerobic glycolysis in cancer.
Maria Cristina Nonato, a professor and researcher at the USP School of Medical Sciences in Ribeirão Preto, is one of the authors of the study and explains that the findings could unfold along various fronts: “Our research group, for example, is especially interested in how proteins from parasites and bacteria interact with human metabolites. This work creates very interesting viewpoints for us to understand the role of different regions on these proteins, not just active sites or their main activities. We need to consider how these proteins interact with other proteins, with other metabolites, the regulatory role in these interactions, etc. There are many possibilities in terms of how metabolic pathways converse and are regulated, and more research is still necessary to understand them.”
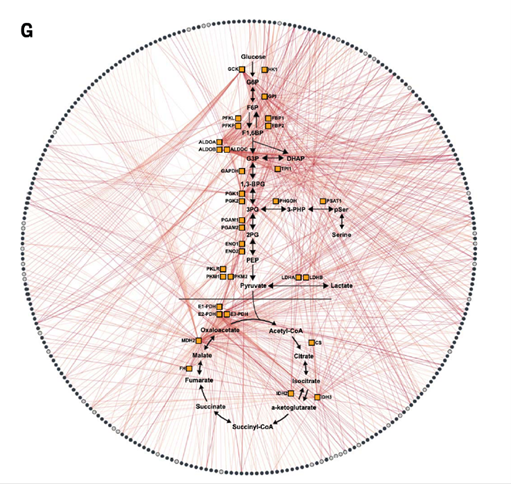
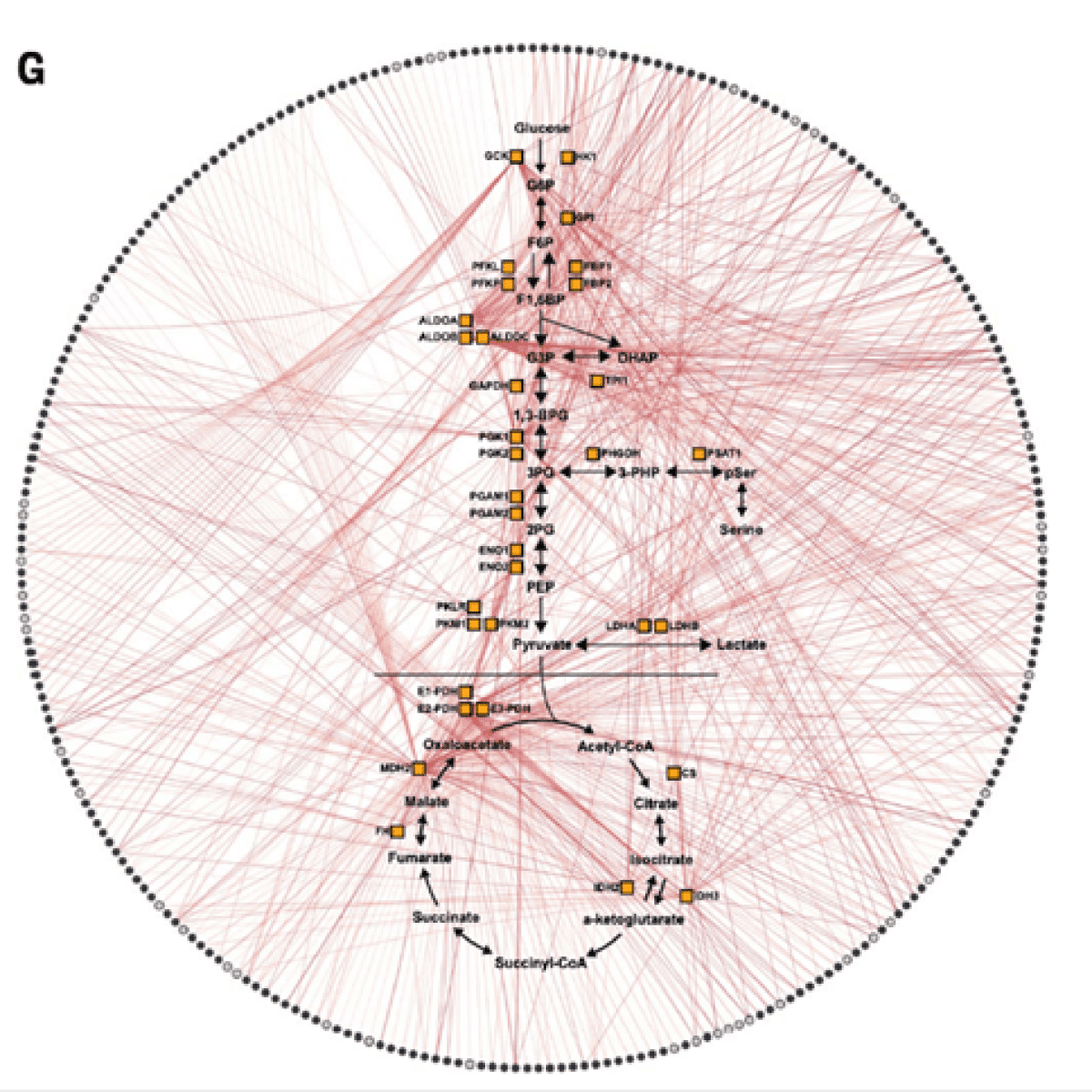
Network of interactions between metabolites (circles comprising the outer edge) and the 33 enzymes in the study. DOI: 10.1126/science.abm3452.
Nonato compares the subtle interactions between the tiny structures to a type of whispered communication. “It is as if these proteins are whispering and sharing information with each other. Understanding this ‘sophisticated conversation’ can reveal important knowledge about the overall behavior of cells.”
Through analyses conducted at Sirius’s Manacá beamline, her group obtained high-resolution measurements of the human protein fumarate hydratase (fumarase), which is involved in a number of metabolic cellular pathways, particularly the tricarboxylic acid cycle. The structure of fumarase was determined in a complex with 2-amino-3-phosphonoproprionic acid, an interaction that had not been previously described. This is a very significant macromolecule for drug development studies; its mutations are known to be associated with several types of cancer.
“This interdisciplinary article combines many techniques and identified hundreds of molecular interactions, but only in some cases were we able to structurally define how the ligand interacts with the protein, which we did using the X-ray diffraction technique at Sirius.”
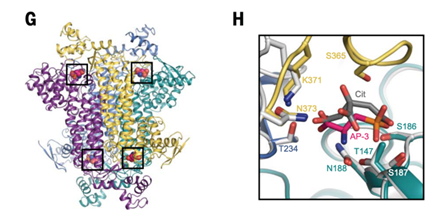
Left: structure of the enzyme fumarate hydratase (FH) with ligands. Right: enlarged view of FH’s active site in interactions with ligands. DOI: 10.1126/science.abm3452.
At Sirius, measurements were also obtained while the beamline was still undergoing scientific commissioning, when experienced researchers test the parameters of the research station. “Manacá is a beamline that is second to none worldwide, not only because of its technical quality but also the support of the team responsible for its operations. Even if you don’t have experience, you can use the beamline and learn a lot from the support team. Everyone works and hopes that the experiment is successful,” adds Nonato.
This publication in Science using the Manacá beamline highlights the rigor of Brazilian science as well as its capacity to overcome frontiers of knowledge using cutting-edge scientific instruments like Sirius, indicating a promising future for experiments conducted with this beamline as well as the other five that are operational and others that will soon be inaugurated, explains Harry Westfahl Jr., Director of the Brazilian Synchrotron Light National Laboratory at the CNPEM.
The library of metabolites has been expanding, and research continues to describe the new structures combined with the binding molecules. Besides human proteins, the researchers are also interested in studying other proteins from parasites, bacteria, and viruses that may be potential targets for new pharmaceuticals, in order to understand how these proteins relate to human metabolites and whether they are affected by them. “The idea is that we can use this methodology, this structure, and this very fruitful collaboration to expand investigations on other fronts,” adds Nonato.
Review article was highlighted in Chemical Reviews magazine
Experiment carried out on Sirius shed light on reaction fundamental to the production of hydrogen fuel