CONTACT & STAFF
For more information on this beamline, contact us.
The MX2 beamline is an experimental station dedicated to Macromolecular Crystallography in the hard x-rays energy range (5 to 15 keV) with maximum flux at 8.5 keV. The station is available for conventional single wavelength data collection as well as for Single and Multiple-wavelength Anomalous Diffraction (SAD/MAD) experiments.
MX2 operates on a 2.0 T hybrid 30-pole wiggler and its optical layout includes collimating mirror, Si(111) double-crystal monochromator and toroidal bendable mirror. The beamline is equipped with PILATUS2M detector from Dectris.
The research projects developed at the MX2 beamline yield precise information about the atomic constitution and shape of macromolecules, such as enzymes and membrane-bound proteins, the building blocks of life inside and outside living cells. This information can lead to a fuller understanding of the biological processes carried by these molecules and by consequence, to the development of new and more efficient medications, cosmetics, biofuels and biomaterials.
Starting from a protein crystal, researchers can obtain information about the three-dimensional shape of the protein, and also run screening studies, where atomic details of small molecules that interact with the protein under study can reveal new avenues for the design of inhibitors. These studies can reveal the detailed mechanisms of the interactions between substrates and enzymes, suggesting improvement or inhibition routes that can guide synthetic or natural products chemists in the development of new commodities.
For more information on this beamline, contact us.
The following experimental techniques and setups are available to users in this beamline. To learn more about the techniques’ limitations and requirements (sample, environment, etc.) contact the beamline coordinator before submitting your proposal.
The station is available for conventional single wavelength data collection as well as for Single and Multiple-wavelength Anomalous Diffraction (SAD/MAD) experiments.
Recent publications using this setup:
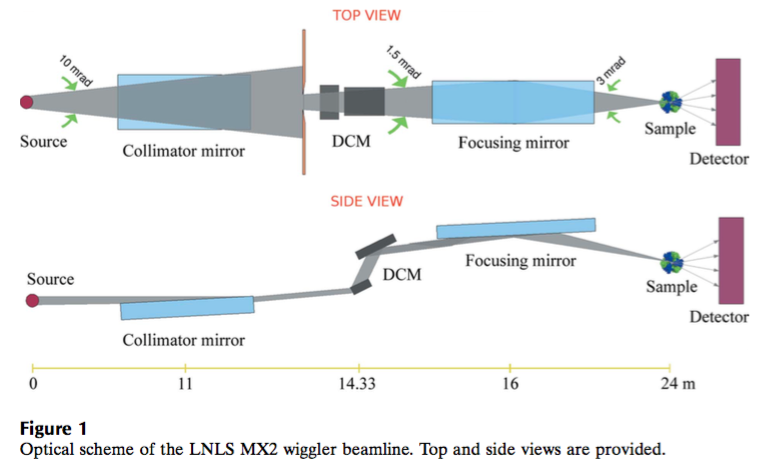
| Element | Type | Position [m] | Description |
|---|---|---|---|
| SOURCE | Insertion Device | 0.00 | Insertion Device W01A, 2.0 T hybrid 30-pole Wiggler, 22 mm minimal gap |
| M1 | Cylindrical Vertical Collimating Mirror | 11.00 | Rh coated ULE glass, θ=4 mrad |
| Mono | Double Crystal Monochromator | 14.26 | Water-cooled Si(111) |
| M2 | Toroidal Horizontal and Vertical Focusing Mirror | 16.00 | Rh coated silicon, θ=4 mrad, mechanical bender |
| Parameter | Value | Condition |
|---|---|---|
| Energy range [keV] | 5-15 | Si(111) |
| Energy resolution [E/E] | 10-4 | Si(111) |
| Beam size at sample [µm2, FWHM] | 150 x 500 | at 8.5 keV |
| Beam divergence at sample [mrad2, FWHM] | 3 | at 8.5 keV |
| Flux density at sample [ph/s/mm2] | 1011 | at 8.5 keV |
| Instrument | Type | Model | Manufacturer | Specifications |
|---|---|---|---|---|
| Detector | Area | PILATUS2M | 172 µm pixel, 1475 x 1679 pixel, 30 kHz frame rate | Dectris |
| Diffractometer | Data collection | MarDTB | Phi and theta rotations. Ionizations chambers, slits, shutter and sample holding. Detector translation range 100 to 350 mm. | Rayonics |
| Cryostats | Nitrogen | Cryojet XL | Sample cooling at 100 Kelvin. | Oxford |
| Detector | Fluorescence | X-123SDD | Si drift diode (SDD). Energy resolution 125 to 140 eV FWHM at 5.9 KeV. Maximum count rate 4 x 106 s-1 Channels number 8. Communication via Ethernet | Amptek |
The optical controls are done through EPICS (Experimental Physics and Industrial Control System), running on a control system called PXI from National Instruments. The experimental control and data collection with the Pilatus 2M detector (Dectris) are done via a RedHat station running the Python based software MXCube. MXCube is the product of an international collaboration for the creation and development of a graphic interface for molecular crystallography beamlines (Gabadinho, J.et al.,2010, J. Synchrotron Rad. 17, 700-707). A mini-kappa goniometer and a robot for in-plate data collection are currently under commissioning.
There are also two machines with RedHat workstation to access one Virtual Machine with the following software applications available for data treatment: iMOSFLM (v1.0.5); HKL2000* (v0.98.695B); CCP4 PACKAGE; SHELX; HKL2MAP (v0.2); PHENIX (v1.7); CNS; COOT; PYMOL; SHARP.
Additional material concerning those software is linked below:
* The HKL-2000 program package available at the beamline is provided on the basis of scientific collaboration free of charge by the program Authors.
The use of the package should be referenced in publication as follows: Z. Otwinowski and W. Minor “Processing of X-ray Diffraction Data Collected in Oscillation Mode”, Methods in Enzymology, Volume 276: Macromolecular Crystallography, part A, p. 307-326, 1997, C.W. Carter, Jr. and R.M. Sweet, Eds., Academic Press.
Researchers who perform non-commercial research may freely use the program for data evaluation and processing while collecting data at the beamline. Researchers from institutions who want to use the program for data evaluation and processing for commercial purposes while collecting data on LNLS beamlines must have an appropriate HKL-2000 license. The license can be obtained from HKL Research, Inc. The HKL-2000 package development is supported by the NIH grant GM-53163 to the laboratories of Drs. Zbyszek Otwinowski (Southwester Medical Center at Dallas) and Wladek Minor (University of Virginia).
Users are required to acknowledge the use of LNLS facilities in any paper, conference presentation, thesis and any other published material that uses data obtained in the execution of their proposal.
GUIMARÃES, B. G.; SANFELICI, L.; NEUENSCHWANDER, R. T.; RODRIGUES, FL.; GRIZOLLI, W. C.; RAULIK, M. A.; PITON, J. R.; MEYER, B. C.; NASCIMENTO, A. S.; POLIKARPOV, I. The MX2 macromolecular crystallography beamline: a wiggler X-ray source at the LNLS. Journal of Synchrotron Radiation, 16: 69-75 (2009). doi:10.1107/S0909049508034870.
The Brazilian Synchrotron Light Laboratory [Laboratório Nacional de Luz Síncrotron (LNLS), Campinas, SP, Brazil] is the first commissioned synchrotron light source in the southern hemisphere. The first wiggler macromolecular crystallography beamline (MX2) at the LNLS has been recently constructed and brought into operation. Here the technical design, experimental set-up, parameters of the beamline and the first experimental results obtained at MX2 are described. The beamline operates on a 2.0 T hybrid 30-pole wiggler, and its optical layout includes collimating mirror, Si(111) double-crystal monochromator and toroidal bendable mirror. The measured flux density at the sample position at 8.7 eV reaches 4.8 x 10^{11} photons s^{-1} mm^{-2} (100 mA)^{-1}. The beamline is equipped with a MarResearch Desktop Beamline Goniostat (MarDTB) and 3 x 3 MarMosaic225 CCD detector, and is controlled by a customized version of the Blu-Ice software. A description of the first X-ray diffraction data sets collected at the MX2 LNLS beamline and used for macromolecular crystal structure solution is also provided.
Scientific publications produced with data obtained at the facilities of this beamline, and published in journals indexed by the Web of Science, are listed below.
Cabral, L. ;Persinoti, G. F.;Paixão, D. A. A.;Martins, M. P. ;Morais, M. A. B.de ;Chinaglia, M.;Domingues, M. N.;Sforça, M. L.;Pirolla, R. A. S. ;Generoso, W. C.;Santos, C. A.;Maciel, L. F. ;Terrapon, N. ;Lombard, V. ;Henrissat, B.;Murakami, M. T.. Gut microbiome of the largest living rodent harbors unprecedented enzymatic systems to degrade plant polysaccharides, Nature Communications, v.13, n.1, p.629, 2022. DOI:10.1038/s41467-022-28310-y
Wegermann, C. A. ;Monzani, E. ;Casella, L. ;Ribeiro, M. A.;Bruzeguini, C. E. T. ;Vilcachagua, J. D. ;Costa, L. A. S. ;Ferreira, A. M. da C.. Unveiling geometrical isomers and tautomers of isatin-hydrazones by NMR spectroscopy, Journal of Molecular Structure, v.1250, n.2, p.131633, 2022. DOI:10.1016/j.molstruc.2021.131633
Fontana, L. A.;Rigolin, V. H.;Ribeiro, M. A.;Barros, W. P.;Megiatto Jr., J. D.. A synthetic tactic to substitute axial ligands in sterically demanding Ru(II)porphyrinates, Dalton Transactions, v.51, p. 9971-9977, 2022. DOI:10.1039/d2dt01095j
Liberato, M. V.;Ramos, B. M. C.;Tomazetto, G.;Crouch, L. I.;Silva, W. J. G. da ;Zeri, A. C. de M.;Bolam, D. N. ;Squina, F. M.. Unique properties of a Dictyostelium discoideum carbohydrate-binding module expand our understanding of CBM–ligand interactions, Journal of Biological Chemistry, v.298, n.5, p.101891, 2022. DOI:10.1016/j.jbc.2022.101891
Cornejo, H. E. L. ;Cenens, W. ;Favaro, D. C.;Sgro, G. G. ;Salinas, R. K.;Guzzo, C. R.;Farah, C. S.. The PilB-PilZ-FimX regulatory complex of the Type IV pilus from Xanthomonas citri, PLoS Pathogens, v.17, n.8, p.e1009808, 2022. DOI:10.1371/journal.ppat.1009808
Gismene, C. ;Hernández González, J. E. ;Santisteban, A. R. N. ;Nascimento, A. F. Z.;Cunha, L. dos S. ;Moraes, F. R. de;Oliveira, C. L. P.;Oliveira, C. C. ;Provazzi, P. J. S. ;Pascutti, P. G.;Arni, R. K.;Mariutti, R. B.. Staphylococcus aureus Exfoliative Toxin E, Oligomeric State and Flip of P186: Implications for Its Action Mechanism, International Journal of Molecular Sciences, v.23, n.17, p.9857, 2022. DOI:10.3390/ijms23179857
Moura, G. T. de; Souza, A. A. ; Garay, A. V.; Freitas, S. M.; Valadares, N. F.. Crystal structure and physicochemical characterization of a phytocystatin from Humulus lupulus: Insights into its domain-swapped dimer, BIOCHIMICA ET BIOPHYSICA ACTA-PROTEINS AND PROTEOMICS, v.1869, n.1,p. 140541, 2021. DOI:10.1016/j.bbapap.2020.140541

Português:
Estação experimental da linha MX2. Da esquerda para a direita: fendas, goniômetro e detector.
English:
MX2 experimental station. From left to right: slits, goniometer and detector.

Português:
Detalhe do goniômetro, beam stopper e cryojet da Linha de Luz MX2.
English:
Details of the goniometer, beam stopper at cryojet at the MX2 beamline.

Português:
Goniômetro e posicionador magnetico de amostras (a ser substituído por um posicionador mini-kappa).
English:
Goniometer and magnetic sample holder (to be replaced by a mini-kappa goniometer).

Português:
Estação experimental da linha MX2 com detector Pilatus2M montado sobre a mesa marDTB.
English:
Experimental station of the MX2 beamline, with Pilatus2M detector mounted over a marDTB bench.

Português:
Goniômetro, Cryojet e Detector Pilatus2M na Linha de Luz MX2.
English:
Goniometer, Cryojet and Pilatus2M Detector at the MX2 beamline.

Português:
Detector Pilatus 2M e mesa de translação.
English:
Pilatus 2M detector and translation table.

Português:
Visão do conjunto experimental, da esquerda para a direita: goniometro, Cryojet, braço robotico (em instalação) e detector Pilatus 2M.
English:
View of experimental setup, from left to right: goniometer, Cryojet, robotic arm (under commissioning) and Pilatus 2M detector.

Português:
Microscópio ótico para seleção de cristais.
English:
Optical microscope for crystal selection.

Português:
Área de controle da estação experimental da Linha de Luz MX2.
English:
Control area of the experimental stations of the MX2 beamline.
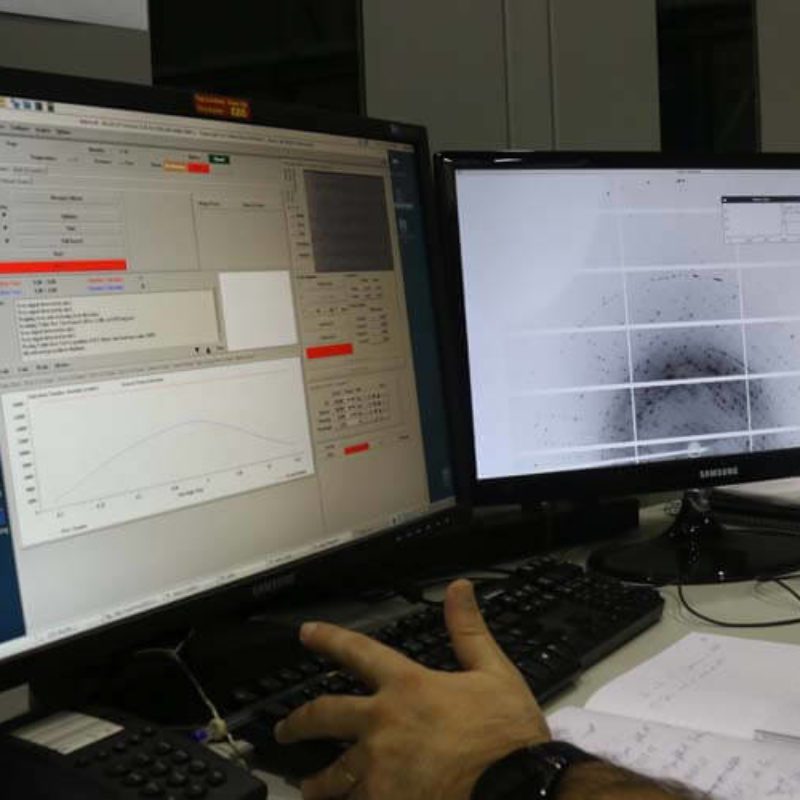
Português:
Interface para coleta de dados na Linha de Luz MX2.
English:
Interface for data collection at the MX2 beamline.