CONTACT & STAFF
For more information on this beamline, contact us.
The SXS beamline is an experimental station dedicated to X-ray Absorption and Photoelectron Spectroscopy in the soft X-rays (1 to 5 keV) energy range. It focuses on to study the electronic, magnetic and geometric structures of materials with applications to atomic and molecular physics, analytical chemistry, environmental and geoscience. Other experimental techniques available include X-ray Magnetic Dichroism and Resonant Auger Spectroscopy.
The SXS beamline is operational for users since 1997 and a broad scientific community including material science, surface science, atomic physics and chemistry among others has used it. Due an increasing demand from the users, in 2009 this beamline had a new X-ray optics in order to provide photons in the energy range from 1000 eV up to 5000 eV.
SXS’ source is a 1.67T bending magnet. There is a nickel-coated and toroidal (1010 x 100 x 100 mm) water cooled mirror that focuses the photon beam at the sample position and it suppresses the harmonic contamination (> 6keV). The incident angle is 0.6 deg and the spot size is 0.6 x 1.2 mm (FWHM) at the sample position.
 The monochromator is a double-crystal with 4 pairs of crystals: Si(111), InSb(111), YB66(400) and Beryl(1010). It works under high vacuum ($5 \times 10^{-8}$ mbar) and the first crystals are maintained below 30 Celsius by a water cooling system.
The monochromator is a double-crystal with 4 pairs of crystals: Si(111), InSb(111), YB66(400) and Beryl(1010). It works under high vacuum ($5 \times 10^{-8}$ mbar) and the first crystals are maintained below 30 Celsius by a water cooling system.
For more information on this beamline, contact us.
The following experimental techniques and setups are available to users in this beamline. To learn more about the techniques’ limitations and requirements (sample, environment, etc.) contact the beamline coordinator before submitting your proposal.
X-ray absorption spectroscopy (XAS) is a widely used technique for determining the local geometric and/or electronic structure of matter.
Setup: Conventional Total electron yield (TEY) and Fluorescence XAS
This setup is optimized for TEY and fluorescence XAS on “standard samples” in standard sample holders. The setup for these experiments, called BioXAS workstation has two electrometers (Io and sample signal), a silicon drift diode (SDD) fluorescence detector, chamber with a differential pumping and a room temperature sample stage (xyzθ). In order to use this setup, samples/environments must fit within our room temperature sample stage.
Recent publications using this setup:
Abdala DB et al., Residence time and pH effects on the bonding configuration of orthophosphate surface complexes at the goethite/water interface as examined by Extended X-ray Absorption Fine Structure (EXAFS) spectroscopy, Journal of Colloid and Interface Science 442 (2015) 15–21;
Andrini L et al., Extended and local structural description of a kaolinitic clay, itsfired ceramics and intermediates: An XRD and XANES analysis, Applied Clay Science 124–125 (2016) 39–45;
Dalfovo MC et al., Real-Time Monitoring Distance Changes in Surfactant-Coated Au Nanoparticle Films upon Volatile Organic Compounds (VOCs), J. Phys. Chem. C (2015), 119, 5098−5106;
Yasser AA et al., Photostability of gold nanoparticles with different shapes: the role of Ag clusters, Nanoscale, 2015, 7, 11273.
Setup: Conventional XPS
This setup is intended for XPS on “standard samples” in standard sample holders. The setup for these experiments, called XPS workstation has two electrometers (Io and sample signal), a hemispherical electron analyzer (Phoibos 150), ultra-high vacuum chamber with base pressure about 5×10-10 mbar and a room temperature motorized sample stage (xyzθ). In order to use this setup, samples/environments must fit within our room temperature sample holder inside a high vacuum pre-chamber. The pre-chamber environment allow submitting the samples to different gas atmospheres, while heating up to 900 °C and, after the treatment, the sample holder is inserted within the analysis chamber, using a load lock system.
Recent publications using this setup:
Garcia-Basabe Y et al., The effect of thermal annealing on the charge transfer dynamics of a donor–acceptor copolymer and fullerene: F8T2 and F8T2:PCBM, Phys.Chem.Chem.Phys., 2015,17, 11244;
Larrude DG et al., Electronic structure and ultrafast charge transfer dynamics of phosphorous doped graphene layers on a copper substrate: a combined spectroscopic study, RSC Adv.,2015,5, 74189;
Martins HP et al., X-ray absorption study of the Fe and Mo valence states in Sr2FeMoO6, Journal of Alloys and Compounds 640 (2015) 511–516;
Silva DO et al., Straightforward synthesis of bimetallic Co/Pt nanoparticles in ionic liquid: atomic rearrangement driven by reduction–sulfidation processes and Fischer–Tropsch catalysis, Nanoscale, 2014, 6, 9085.
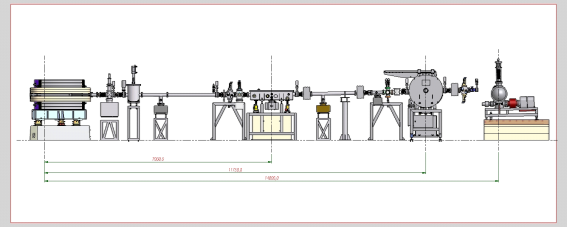
| Element | Type | Position [m] | Description |
|---|---|---|---|
| SOURCE | Bending Magnet | 0.00 | Bending Magnet D04 exit A (4°), 1.67 T |
| Mirror | Toroidal Horizontal and Vertical Focusing Mirror | 7.00 | Ni coated, RT = 668m, RS = 73mm, θ = 10 mrad |
| Mono | Double Crystal Monochromator | 11.75 | Water-cooled InSb(111), Si(111), YB66 (400) and Beryl(10-10) |
| Parameter | Value | Condition |
|---|---|---|
| Energy range [keV] | 1-5 | Si(111) |
| Energy resolution [ΔE/E] | 10-4 | Si(111) |
| Beam size at sample [mm2, FWHM] | 0.6 x 1.2 | at 3 keV |
| Beam divergence at sample [mrad2, FWHM] | 0.2 x 4 | at 3 keV |
| Flux density at sample [ph/s/mm2] | 4 x 1011 | at 3 keV |
| Instrument | Type | Model | Manufacturer | Specifications |
|---|---|---|---|---|
| Total electron yield detector | Electrometer | 6514 | 20 pA – 2 mA range | Keithley |
| Fluorescence Detector | Silicon drift diode | SuperFast SDD | Area: 25 mm2; Energy resolution: 125–155 eV | Amptek |
| Photoelectron analyzer | Hemispherical | Phoibos 150 | MCD with 9 channels; 3500 eV kinetic energy | Specs |
| Furnace | Halogen lamps/ high vacuum | – | Max Temp.: 900°C | LNLS in-house development |
| Sputtering | Argon | IG2 ion source | 0.5-2kV; 2.5mm beam diameter at 25 mm | RBD |
| Residual gas analyzer | Mass spectrometer | RGA200 | 1-200 amu; quadrupole | SRS |
| Sample charge neutralizer | Electron flood gun | FG15/40 | 0-500 eV; 0-5 mA | Specs |
| Sample Cells | Liquid | – | Ultralene window; 4 x 113 µL | LNLS in-house development |
All beamline controls are done through EPICS (Experimental Physics and Industrial Control System), running on a PXI from National Instruments. The data acquisition is done using a Red Hat workstation with the Py4Syn, developed at LNLS by SOL group. CSS (Control System Studio) is used as a graphical interface to display and control the beamline devices.
Users are required to acknowledge the use of LNLS facilities in any paper, conference presentation, thesis and any other published material that uses data obtained in the execution of their proposal.
ABBATE, F. C. VICENTIN, V. COMPAGNON-CAILHOL, M. C. ROCHA AND H. TOLENTINO, The soft X-ray spectroscopy beamline at the LNLS: technical description and commissioning results, J. Synchrotron Rad., 6, 964 (1999). doi:10.1107/S0909049599008122.
The soft X-ray spectroscopy beamline installed at a bending-magnet source in the LNLS is presented. A technical description of the main elements is given and some selected commissioning results are shown. The beamline optics was designed to cover the soft X-ray energy range from 790 up to 4000 eV. The bending-magnet source has a critical energy of 2.08 keV and delivers ~10^{12} photons.s^{-1}.mradH^{-1} (0.1% bandwidth)^{-1}(100 mA)^{-1}. The focusing element is a gold-coated toroidal mirror operating at an angle of incidence of 1°. The double-crystal monochromator has three pairs of crystals which can be selected by a lateral translation. The UHV experimental station is equipped with an ion gun, an electron gun, a LEED optics and an electron analyzer. The beamline is intended for X-ray absorption, photoemission, reflectivity and dichroism experiments. The beamline has been installed, commissioned, and is now open to the external users community.
TOLENTINO, V. COMPAGNON-CAILHOL, F. C. VICENTIN AND M. ABBATE,The LNLS soft X-ray spectroscopy beamline, J. Synchrotron Rad., 5, 539 (1998). doi:10.1107/S0909049597016087.
The soft X-ray spectroscopy beamline installed at a bending-magnet source at the LNLS is described. The optics are designed to cover energies from 800 to 4000 eV with good efficiency. The focusing element is a gold-coated toroidal mirror with an angle of incidence of 17 mrad. The UHV double-crystal monochromator has three pairs of crystals, Si (111), InSb (111) and beryl (10-10), that can be selected by a sliding movement. The UHV workstation is equipped with an ion gun, an electron gun, an electron analyser, LEED optics, an open channeltron and a photodiode array. This beamline is intended for photoemission, photoabsorption, reflectivity and dichroism experiments.
Scientific publications produced with data obtained at the facilities of this beamline, and published in journals indexed by the Web of Science, are listed below.
Firmano, R. F. ;Colzato, M.;Alleoni, L. R. F.. Phosphorus speciation and distribution in a variable-charge Oxisol under no-till amended with lime and/or phosphogypsum for 18 years, European Journal of Soil Science, v.73, n.1, p.e13198, 2022. DOI:10.1111/ejss.13198
Costa, R. F. ;Firmano, R. F. ;Colzato, M.;Crusciol, C. A. C. ;Alleoni, L. R. F.. Sulfur speciation in a tropical soil amended with lime and phosphogypsum under long-term no-tillage system, Geoderma, v.406, p.115461, 2022. DOI:10.1016/j.geoderma.2021.115461
Richard, D. ;Martínez, J. ;Mizrahi, M.;Andrini, L. R.;Rendtorff, N. M.. Assessment of structural order indices in kaolinites: A multi-technique study including EXAFS, Journal of Electron Spectroscopy and Related Phenomena, v.254, p.147128, 2022. DOI:10.1016/j.elspec.2021.147128
Calvo, A.;Andrini, L. R.;Williams, F. J.;Ramallo-López, J. M.;Soler-Illia, G. J. A. A.;Requejo, F. G.. Controlling the local-ensemble structure in mesoporous hybrid titania-silica thin films containing aminopropyl groups, Journal of Sol-Gel Science and Technology, v.102, p.172–184 , 2022. DOI:10.1007/s10971-021-05579-x
Porto, V. ;Buceta, D.;Domínguez, B. ;Carneiro, C. ;Borrajo, E. ;Fraile, M.;Davila-Ferreira, D. ;Arias, I. R. ;Blanco, J. M.;Blanco, M. C.;Devida, J. M. ;Giovanetti, L. J.;Requejo, F. G.;Hernández-Garrido, J. C. ;Calvino, J. J.;López-Haro, M.;Barone, G.;James, A. M. ;García-Caballero, T.;González-Castaño, D. M. ;Treder, M. ;Huber, W. ;Vidal, A. ;Murphy, M. P. ;López-Quintela, M. A.;Domínguez, F.. Silver Clusters of Five Atoms as Highly Selective Antitumoral Agents Through Irreversible Oxidation of Thiols, Advanced Functional Materials, v.32, n.29, p.2113028, 2022. DOI:10.1002/adfm.202113028
Tucker, C. L. ;Ragoo, Y. ;Mathe, S. ;Macheli, L.;Bordoloi, A. ;Rocha, T. C. R.;Govender, S.;Kooyman, P. J.;Van Steen, E.. Manganese promotion of a cobalt Fischer-Tropsch catalyst to improve operation at high conversion, Journal of Catalysis, v.411, p.97-108, 2022. DOI:10.1016/j.jcat.2022.05.006
Misael, W. A. ;Péan, E. V. ;Borges, B. G. A. L.;Mello, G. da C. ;Wouk, L. ;Davies, M. L.;Roman, L. S.;Pereira, M. L. R. D.. Molecular Orientation and Femtosecond Electron Transfer Dynamics in Halogenated and Nonhalogenated, Eco-Friendly Processed PTB7- Th, ITIC, PTB7-Th:ITIC, and PTB7-Th:PCBM Films, Journal of Physical Chemistry C, v.126, n.26, p.10807–10817, 2022. DOI:10.1021/acs.jpcc.2c01298

Português:
Cabana Óptica com vista para câmara do monocromador.
English:
Optical hutch showing the monochromator chamber.

Português:
Cabana Óptica com vista para a câmara do espelho.
English:
Optical hutch showing the mirror chamber.

Português:
Estação de Espectroscopia por Fotoelétrons (XPS) acoplada à Linha de Luz SXS.
English:
X-ray Photoelectron Spectroscopy (XPS) workstation coupled to the SXS beamline.

Português:
Janela de exibição do porta-amostra da estação de Espectroscopia por Fotoelétrons.
English:
Sample holder viewport at the X-ray Photoelectron Spectroscopy (XPS) workstation.

Português:
Estação de trabalho de Espectroscopia por Fotoelétrons (XPS) mostrando a câmara principal, pré-câmara (utilizada para o tratamento e transferência de amostras), manipulador motorizado, bomba de titânio para sublimação, analisador de elétrons, eletrômetros e equipamentos acessórios.
English:
X-ray Photoelectron Spectroscopy (XPS) workstation showing the main chamber, pre-chamber (used for sample treatment and transfer), motorized manipulator, titanium sublimation pump, electron analyzer, electrometers and accessory equipments.

Português:
Estação de Espectroscopia por Fotoelétrons (XPS) mostrando a transferência de amostras.
English:
X-ray Photoelectron Spectroscopy (XPS) workstation showing sample transfer.

Português:
Visão geral da Linha de Luz SXS durante experimento com usuários externos.
English:
SXS beamline overview during an experiment with external users.

Português:
Visão geral da Linha de Luz SXS, com vista para a eletrônica de controle, cabana óptica e estação de trabalho.
English:
SXS beamline overview, showing control electronics, optical hutch and workstation.