
Equipment improves the investigation of materials for fuel cells, batteries and electrolysers
Fossil fuels are the main source of energy in the world. However, the search for clean, renewable, and cheap energy sources has intensified recently, especially with the growing consensus that the rise in the average temperature of the planet is caused by human action. In this context, electrochemical devices, which involve reactions for the transformation of chemical energy into electrical energy, appear as a viable option to fossil fuels.
Among those available are fuel cells and batteries, capable of converting the chemical energy of molecules into electrical energy and storing it, and electrolysers capable of converting low-cost molecules into more economically attractive molecules. Thus, to improve the performance of these electrochemical devices, it is essential to understand the processes that occur between their components, more precisely in the interaction between the electrodes and the electrolyte.
For this reason, researchers from the State University of Campinas (UNICAMP), in collaboration with researchers from the Brazilian Center for Research in Energy and Materials (CNPEM) and the Federal University of São Carlos (UFSCar), developed an electrochemical cell [1] with the objective to perform various types of in situ experiments. These experiments allow direct access to the dynamics of electrochemical reactions in real time and make it possible to understand the processes that occur in the system from an atomic and molecular point of view. Hence, it is possible to optimize the materials that are part of fuel cells, batteries and electrolysers mentioned, and also of devices such as supercapacitors and electrochemical sensors, among others.
The cell developed by the group, shown in Figure 1, is a versatile electrochemical cell, which can be used in experiments involving several techniques, such as in situ Raman spectroscopy, in situ infrared spectroscopy, X-ray absorption and diffraction, among others, in addition to being compatible with the electrochemical response obtained in laboratory cells. Thus, it is possible to perform all types of material characterization by different experimental techniques using a single electrochemical cell.
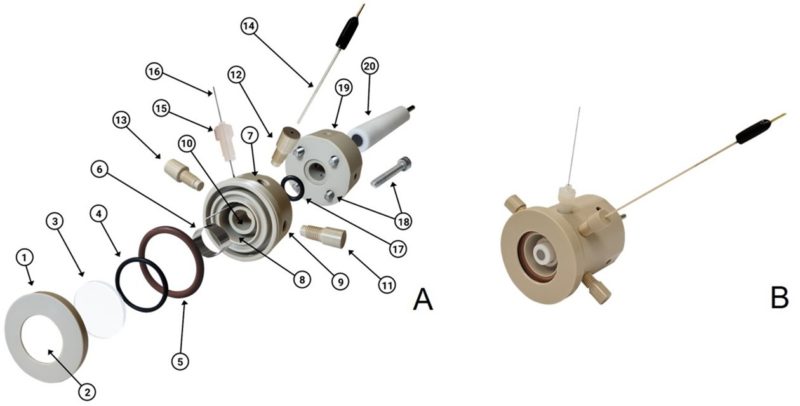
Figure 1: A, B) Schematic drawings of the SEC: threaded lip (1); aperture for passing the radiation beam and, in the case of a photoelectrochemical experiment, to illuminate the electrode with a solar simulator or LEDs (2); window (3); O-rings (4, 5, 17); CE (6 16); SEC body – part 1 (7); chamber for the electrolyte, the CE and the RE (8); electrolyte inlet and outlet (9, 11, 13), WE inlet (10); RE inlet (12); RE (14); CE inlet (15); bolt (18); SEC body – part 2 (19); WE (20).
This versatile electrochemical cell even allows the use of synchrotron light techniques under the same conditions of laboratory electrochemical characterization, which is of particular importance in the context of the new Brazilian synchrotron light source, Sirius, which will improve the characterization capacity of electrochemical processes and samples under in situ operating conditions.
The published work was on the cover of the journal ChemElectroChem (2020).
Source: [1] J. L. Bott-Neto, M. V. F. Rodrigues, M. C. Silva, E. B. Carneiro-Neto, G. Wosiak, J. C. Mauricio, E. C. Pereira, S. J. A. Figueroa, P. S. Fernández, Versatile Spectroelectrochemical Cell for In Situ Experiments: Development, Applications, and Electrochemical Behavior, ChemElectroChem 2020, 7, 4306. DOI: 10.1002/celc.202001242
Research reveals mechanisms of action of iron nanoparticles used in aquifer decontamination
Research explores wave-particle duality to accelerate light in a two-dimensional crystal using functional substrates